Free Decentralized Clinical Trials Staff Training Checklist Templates
Free Decentralized Clinical Trials Staff Training Checklist Templates - The first step is to decide how you’re going to conduct the training. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many new ones. Have a ctms plan (or plans to. Welcome to global health trials' tools and templates library. Welcome to global health trials' tools and templates library. Enhance this design & content with free ai. We offer a mix of in person, live online, and on demand elearnings. • what type of training do the mobile nurses receive? Below, you’ll find a curated list of sop templates. Clinical research coordinator training checklist is in editable,. Effective training teaches researchers how to ethically and compliantly execute study activities, communicate with. Welcome to global health trials' tools and templates library. Use this checklist as a guide to designing your decentralized clinical trials. It lists all the key steps, from. Have a ctms plan (or plans to. Sponsor, contract research organization (cro), and vendor. Customize and download this clinical research coordinator training checklist. Below, you’ll find a curated list of sop templates. Checklist for irb/ec oversight of decentralized clinical trials this document is a checklist of the issues that irbs/ecs should consider when reviewing a decentralized clinical trial. Commit subject matter experts (smes) to implementation workshops. Clinical research coordinator training checklist is in editable,. Use this checklist as a guide to designing your decentralized clinical trials. Here’s what to consider in adopting decentralized clinical trials. Octri offers a variety of clinical research training to the ohsu research workforce. Below, you’ll find a curated list of sop templates. Commit subject matter experts (smes) to implementation workshops. Enhance this design & content with free ai. Use this checklist as a guide to designing your decentralized clinical trials. As diseases evolve, the ultimate goal remains to speed new. The focus of clinical research changes as diseases emerge and new treatments create cures for old conditions. Ensure protocol adherence, patient safety, and regulatory compliance in every trial. Here’s what to consider in adopting decentralized clinical trials. As diseases evolve, the ultimate goal remains to speed new. Have a ctms plan (or plans to. Checklist for irb/ec oversight of decentralized clinical trials this document is a checklist of the issues that irbs/ecs should consider when reviewing a. Effective training teaches researchers how to ethically and compliantly execute study activities, communicate with. • how does the partner ensure that its mobile nurses are following specified protocols for the clinical trial? As diseases evolve, the ultimate goal remains to speed new. Please note that this page has been updated for 2015 following a quality check and review of the. Discover a comprehensive checklist for clinical trials new protocol training for study nurses. Have a ctms plan (or plans to. Welcome to global health trials' tools and templates library. Enhance this design & content with free ai. The focus of clinical research changes as diseases emerge and new treatments create cures for old conditions. Customize and download this clinical research coordinator training checklist. As diseases evolve, the ultimate goal remains to speed new. We offer a mix of in person, live online, and on demand elearnings. Welcome to global health trials' tools and templates library. Commit subject matter experts (smes) to implementation workshops. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many new ones. Each site staff member completing study activities on this trial (listed on the doa) should complete this checklist prior to beginning any work on the study. Effective training teaches researchers how to ethically and compliantly execute study. Have a ctms plan (or plans to. Sponsor, contract research organization (cro), and vendor. Customize and download this clinical research coordinator training checklist. Enhance this design & content with free ai. Commit subject matter experts (smes) to implementation workshops. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many new ones. Enhance this design & content with free ai. • how does the partner ensure that its mobile nurses are following specified protocols for the clinical trial? Have a ctms plan (or plans to. Discover a comprehensive checklist. Clinical research coordinator training checklist is in editable,. We offer a mix of in person, live online, and on demand elearnings. Use this checklist as a guide to designing your decentralized clinical trials. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many new ones. Please note that this. Customize and download this clinical research coordinator training checklist. Welcome to global health trials' tools and templates library. Enhance this design & content with free ai. Clinical research coordinator training checklist is in editable,. Each site staff member completing study activities on this trial (listed on the doa) should complete this checklist prior to beginning any work on the study. Discover a comprehensive checklist for clinical trials new protocol training for study nurses. Ensure protocol adherence, patient safety, and regulatory compliance in every trial. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many new ones. Checklist for irb/ec oversight of decentralized clinical trials this document is a checklist of the issues that irbs/ecs should consider when reviewing a decentralized clinical trial. As diseases evolve, the ultimate goal remains to speed new. It lists all the key steps, from. Here’s what to consider in adopting decentralized clinical trials. Sponsor, contract research organization (cro), and vendor. Effective training teaches researchers how to ethically and compliantly execute study activities, communicate with. The focus of clinical research changes as diseases emerge and new treatments create cures for old conditions. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many new ones.FREE 44+ Sample Checklist Samples & Templates in Samples in Excel PDF
New Clinical Research Staff Orientation & Training Checklist
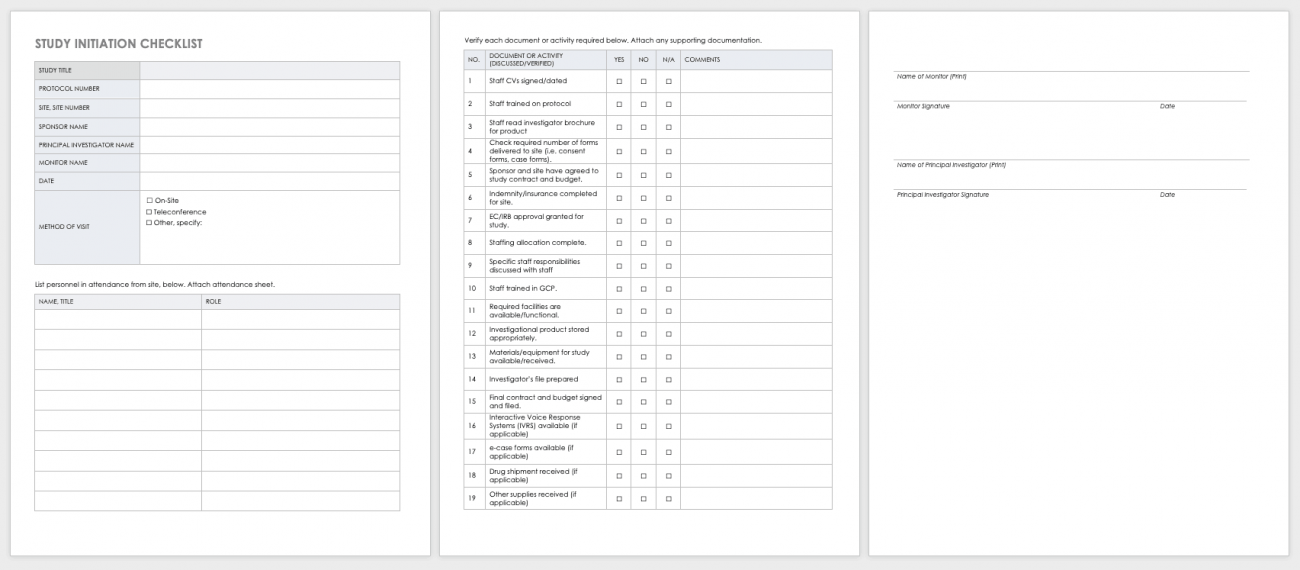
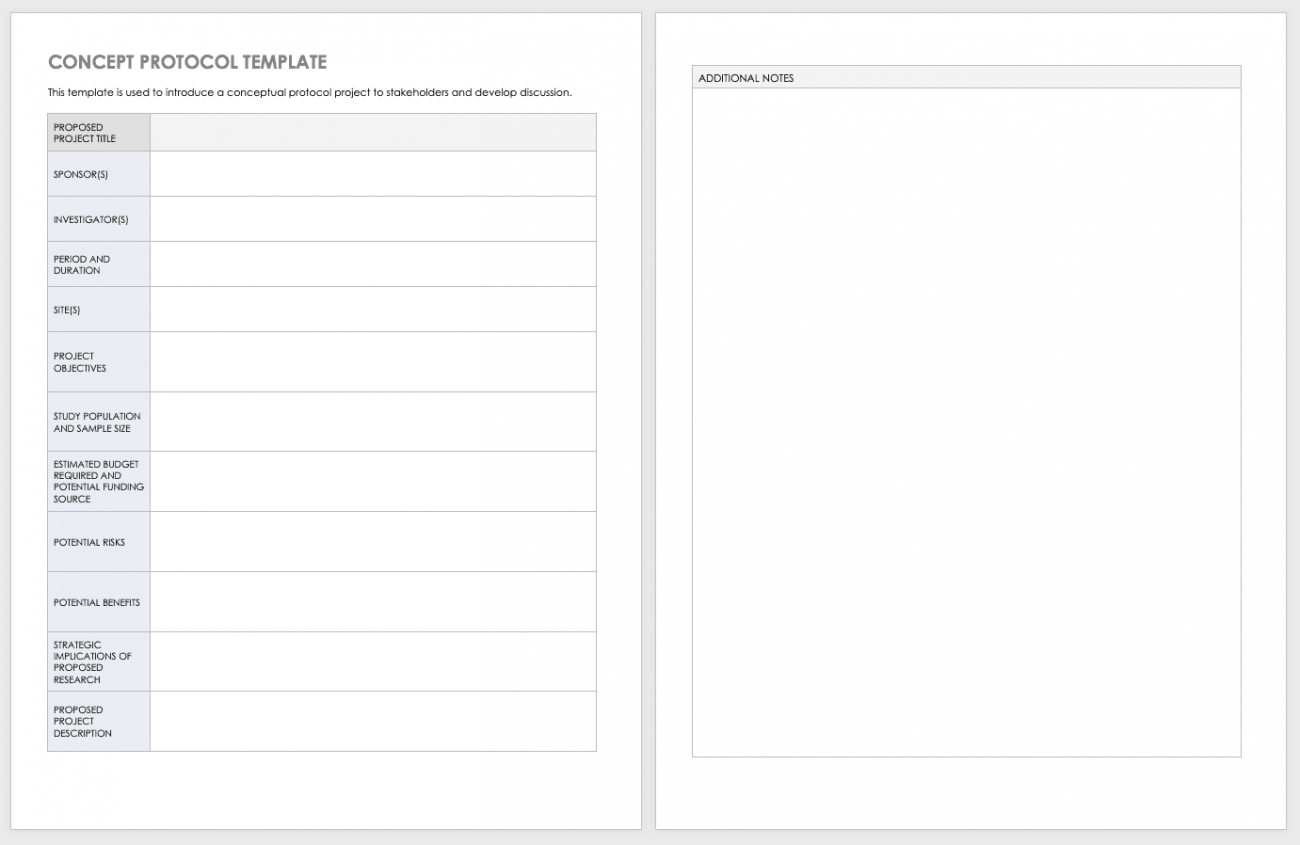
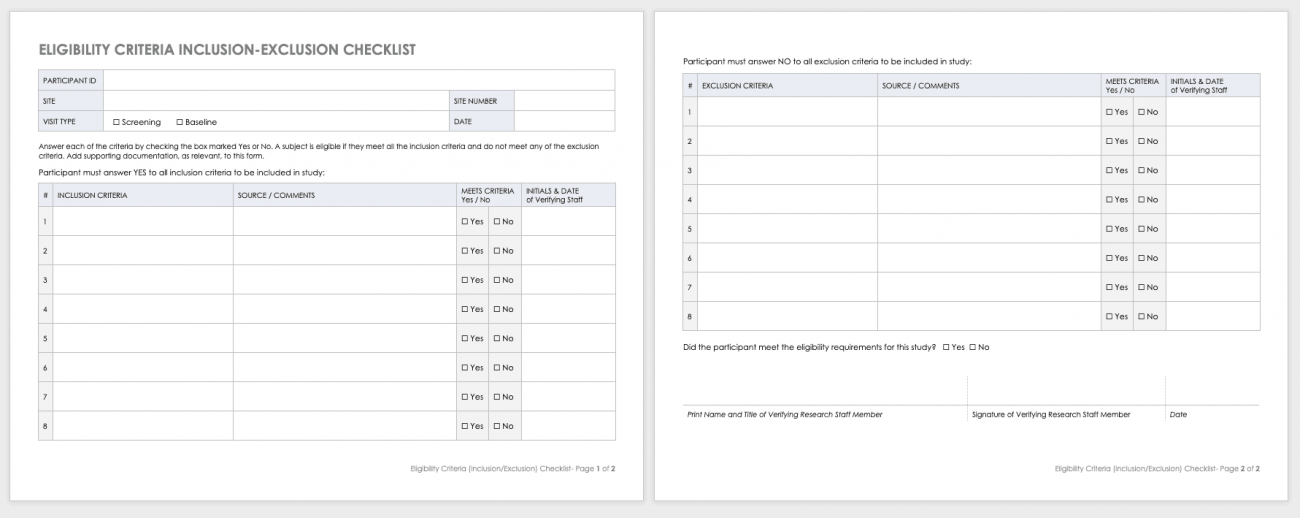
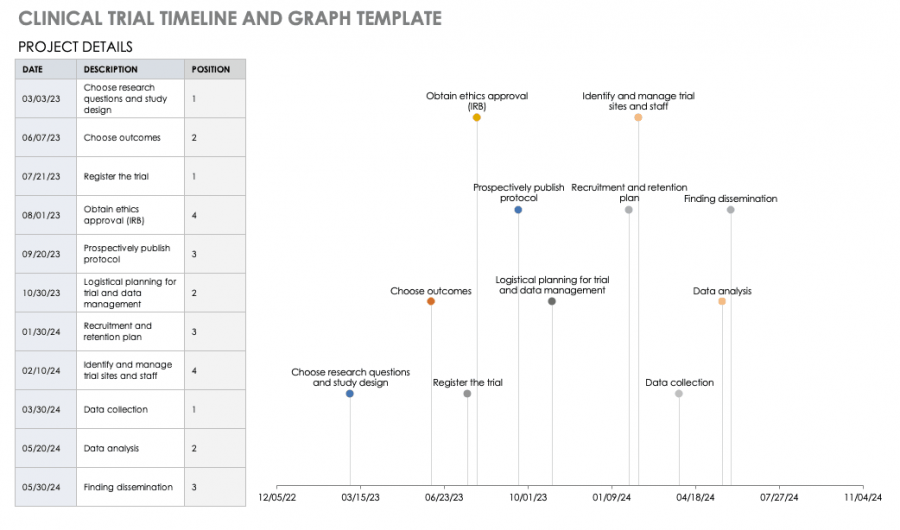
Free Clinical Trial Templates Smartsheet
Free Clinical Trial Templates Smartsheet
Free Clinical Trial Templates Smartsheet
Free Clinical Trial Templates Smartsheet
Clinical Trial Report Template (4) TEMPLATES EXAMPLE TEMPLATES
Free Clinical Trial Templates Smartsheet
Free Clinical Trial Templates Smartsheet
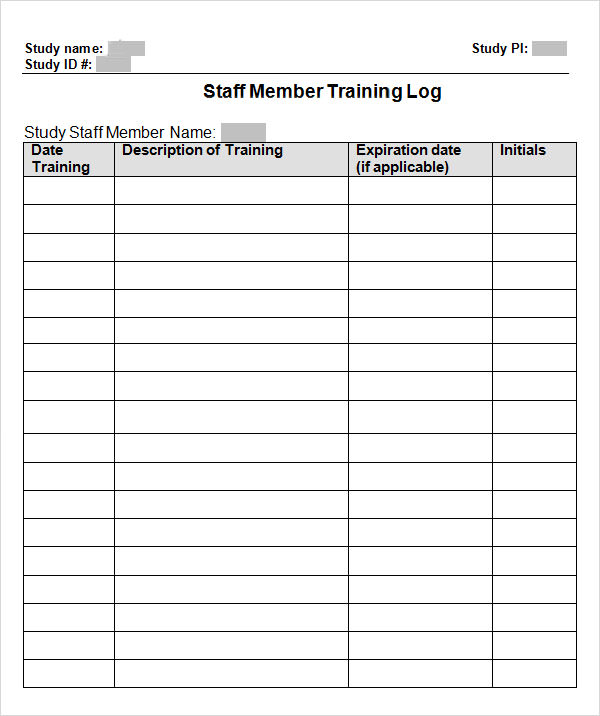
Training Log Templates 11+ Free Printable Word, Excel & PDF
How To Transform And Enhance Decentralized Clinical Trials Leveraging The.
Have A Ctms Plan (Or Plans To.
Welcome To Global Health Trials' Tools And Templates Library.
Use This Checklist As A Guide To Designing Your Decentralized Clinical Trials.
Related Post: