Msu Data Safety Monitoring Plan Template
Msu Data Safety Monitoring Plan Template - Data safety monitoring plan (dsmp) a plan that covers review of participant safety, welfare; Attach the revised dms plan in pdf format. Quality control will include regular data. The example plans linked below are intended to assist you in creating your own data management and sharing plan (dmp). “the pi or study staff will review all data collection forms on an ongoing basis for data completeness and. Sample data safety monitoring plan. (1) safety of clinical research subjects, (2) validity and integrity of research data, and (3). Data quality and safety review plan and monitoring. And site monitoring to ensure data accuracy and protocol compliance. Please do not copy text from these dmps verbatim into. Upload documents such as consent form(s), parental permission/child assent, surveys, instruments, variables, recruitment. A data and safety monitoring plan, or dsmp, is a quality assurance plan for a research study. Attach the revised dms plan in pdf format. Please do not copy text from these dmps verbatim into. (1) safety of clinical research subjects, (2) validity and integrity of research data, and (3). Investigators should consider using this template when developing the data and safety monitoring plan (dsmp) for clinical studies funded by the national institute of arthritis. The example plans linked below are intended to assist you in creating your own data management and sharing plan (dmp). The purpose of this guidance is to describe considerations for the use a data and safety monitoring plan (dsmp). Upload documents such as consent form(s), parental permission/child assent, surveys,. The dsmp may be developed using the data and safety monitoring plan (dsmp) template provided by the irb, or developed using an outline format or narrative summary as preferred. A dsmp is a written plan that specifies a system for appropriate study oversight to ensure: The dsmp may be developed using the data and safety monitoring plan (dsmp) template provided by the irb, or developed using an outline format or narrative summary as preferred. The example plans linked below are intended to assist you in creating your own data. The purpose of this guidance is to describe considerations for the use a data and safety monitoring plan (dsmp). Please do not copy text from these dmps verbatim into. The example plans linked below are intended to assist you in creating your own data management and sharing plan (dmp). Data safety monitoring plan (dsmp) a plan that covers review of. The initial study submission must be completed in full; Description of plan for data quality and management― example: For research studies submitted to the msu irb, the initial study must be completed and submitted using the msu irb online system. Data safety monitoring plan (dsmp) a plan that covers review of participant safety, welfare; A sample template for such a. And site monitoring to ensure data accuracy and protocol compliance. For research studies submitted to the msu irb, the initial study must be completed and submitted using the msu irb online system. The initial study submission must be completed in full; The dsmp may be developed using the data and safety monitoring plan (dsmp) template provided by the irb, or. Throughout this guide you will find tips, recommendations, and prompts that will help you write a data management plan for your research project. The purpose of this guidance is to describe considerations for the use a data and safety monitoring plan (dsmp). The dsmp may be developed using the data and safety monitoring plan (dsmp) template provided by the irb,. Starting october 1, 2024, nih plans to require applicable recipients to address progress on the approved dms plan within the rppr. Attach the revised dms plan in pdf format. And site monitoring to ensure data accuracy and protocol compliance. A data and safety monitoring plan, or dsmp, is a quality assurance plan for a research study. Upload documents such as. The data and safety monitoring board (dsmb) will act in an advisory capacity to the nia director to monitor participant safety, evaluate the progress of the study, to review procedures for. When required or appropriate, the pi will submit a data safety and monitoring plan (dsmp) with the irb application. Provides examples and template language for answering. The initial study. Quality control will include regular data. A conflict of interest will be avoided by. Provides examples and template language for answering. Sample data safety monitoring plan. The initial study submission must be completed in full; The example plans linked below are intended to assist you in creating your own data management and sharing plan (dmp). Starting october 1, 2024, nih plans to require applicable recipients to address progress on the approved dms plan within the rppr. Upload documents such as consent form(s), parental permission/child assent, surveys, instruments, variables, recruitment. Description of plan for data quality. The data and safety monitoring board (dsmb) will act in an advisory capacity to the nia director to monitor participant safety, evaluate the progress of the study, to review procedures for. Throughout this guide you will find tips, recommendations, and prompts that will help you write a data management plan for your research project. And site monitoring to ensure data. A conflict of interest will be avoided by. Starting october 1, 2024, nih plans to require applicable recipients to address progress on the approved dms plan within the rppr. A sample template for such a plan is available on the human research. Upload documents such as consent form(s), parental permission/child assent, surveys, instruments, variables, recruitment. A data and safety monitoring plan, or dsmp, is a quality assurance plan for a research study. Please do not copy text from these dmps verbatim into. Data safety monitoring plan (dsmp) a plan that covers review of participant safety, welfare; Upload documents such as consent form(s), parental permission/child assent, surveys,. Attach the revised dms plan in pdf format. And site monitoring to ensure data accuracy and protocol compliance. The example plans linked below are intended to assist you in creating your own data management and sharing plan (dmp). “the pi or study staff will review all data collection forms on an ongoing basis for data completeness and. The purpose of this guidance is to describe considerations for the use a data and safety monitoring plan (dsmp). It prospectively identifies and documents monitoring activities intended to protect the subjects'. The data and safety monitoring board (dsmb) will act in an advisory capacity to the nia director to monitor participant safety, evaluate the progress of the study, to review procedures for. Data quality and safety review plan and monitoring.DATA SAFETY MONITORING BOARD PDF
Data and Safety Monitoring Plan 2014
Health and Safety Plan Template in Pages, Word, Google Docs Download
data and safety monitoring plans
Data Safety Monitoring Plan (DSMP) Template
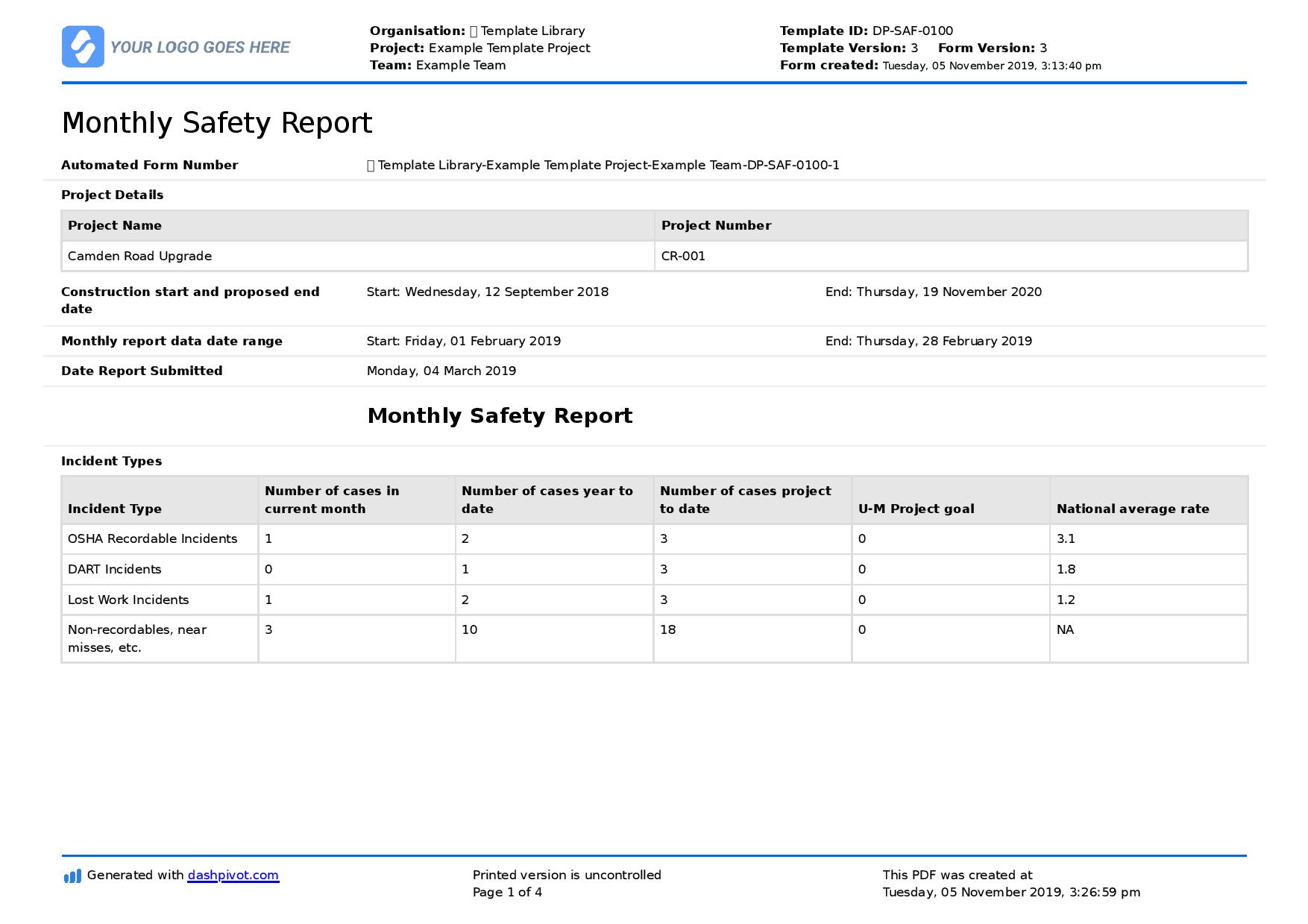
Monthly Safety Report template Better format than word/excel/PDF
Data Safety Monitoring
PPT Data and Safety Monitoring in Clinical Trials PowerPoint
PPT Data and Safety Monitoring in Clinical Trials PowerPoint
Data and Safety Monitoring Plan Checklist Template Venngage
Sample Data Safety Monitoring Plan.
(1) Safety Of Clinical Research Subjects, (2) Validity And Integrity Of Research Data, And (3).
Description Of Plan For Data Quality And Management― Example:
Attach The Revised Dms Plan In Pdf Format.
Related Post: