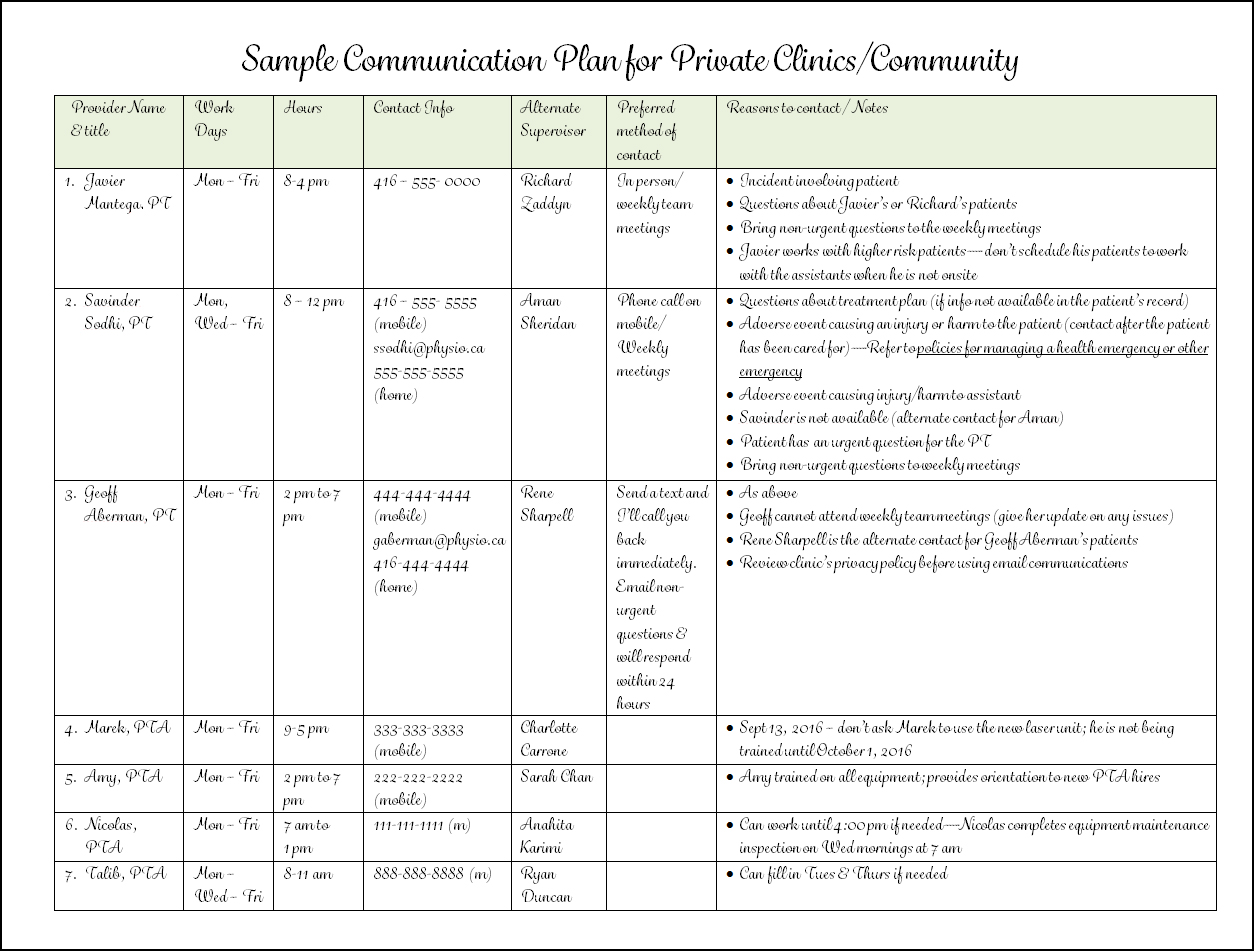
Transcelerate Protocol Template
Transcelerate Protocol Template - In november 2018, transcelerate released its first version of a csr template. The transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration clinical trials protocol. Transcelerate protocol is in editable, printable format. In a blog post announcing the new template, peter marks, the director of the. The common protocol template (cpt), published by transcelerate harmonizes and streamlines the content of clinical trial protocols. Resources for our new common protocol template is now accessible via download. Once the form is completed, please click the submit button. Clinical content & reuse (cc&r) initiative, formerly known as common protocol template (cpt), enhanced clinical trial processes by developing common content for reuse across. This update to the cpt. Reinforces need for harmonized protocol formats and supports use of data standards for required trial objectives and endpoints. In a blog post announcing the new template, peter marks, the director of the. Transcelerate, a nonprofit organization, has a similar common protocol template (cpt) under development. We provide support in adopting and implementing transcelerate common protocol template for better clinical trial protocol standardization. Reinforces need for harmonized protocol formats and supports use of data standards for required trial objectives and endpoints. The main goal achieved by the template is to facilitate the. Once the form is completed, please click the submit button. This video explores out common protocol template (cpt) initiative and highlights how a harmonized cpt can simplify trial protocols, regulatory reviews and benefit multiple stakeholders. A master protocol template provides guidance for the key components needed to conduct and analyse platform trials. In november 2018, transcelerate released its first version of a csr template. Clinical content & reuse (cc&r) initiative, formerly known as common protocol template (cpt), enhanced clinical trial processes by developing common content for reuse across. The main goal achieved by the template is to facilitate the. Resources for our new common protocol template is now accessible via download. Plus, resources to support their use, implementation, and adoption. Templates for the common protocol (cpt), statistical analysis plan (sap), and clinical study report (csr) are available here. This video explores out common protocol template (cpt) initiative and. Please fill out the form below to download the common protocol template and other materials. The transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration clinical. Customize and download this transcelerate protocol. The transcelerate common protocol template (cpt) core structure has been aligned with the us national. Customize and download this transcelerate protocol. Clinical content & reuse (cc&r) initiative, formerly known as common protocol template (cpt), enhanced clinical trial processes by developing common content for reuse across. Plus, resources to support their use, implementation, and adoption. Clinical template suite (cts);template, resources, and use guidance. In november 2018, transcelerate released its first version of a csr template. Transcelerate has been developing a protocol template for a while, recently launching the common protocol template that has been harmonized with proposed model. The transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration clinical. This csr template and associated resources, including a template for statistical analysis plans. The main goal achieved by the template is to facilitate the. Today announced the availability of an enhanced technology enabled common protocol template (cpt). We provide support in adopting and implementing transcelerate common protocol template for better clinical trial protocol standardization. Clinical template suite (cts);template, resources, and use guidance. Transcelerate protocol is in editable, printable format. Plus, resources to support their use, implementation, and adoption. Today announced the availability of an enhanced technology enabled common protocol template (cpt). Clinical template suite (cts);template, resources, and use guidance. The transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration clinical trials protocol. Once the form is. The main goal achieved by the template is to facilitate the. This csr template and associated resources, including a template for statistical analysis plans (saps),. Clinical template suite (cts);template, resources, and use guidance. Within the next two years, ich is expected to issue a guideline, template and technical specification for a common electronic structured protocol, built in part on the.. This update to the cpt. Enhance this design & content with free ai. The transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration clinical trials protocol. Within the next two years, ich is expected to issue a guideline, template and technical specification for a common electronic structured. Templates for the common protocol (cpt), statistical analysis plan (sap), and clinical study report (csr) are available here. Customize and download this transcelerate protocol. Once the form is completed, please click the submit button. Resources for our new common protocol template is now accessible via download. In november 2018, transcelerate released its first version of a csr template. Templates for the common protocol (cpt), statistical analysis plan (sap), and clinical study report (csr) are available here. Clinical content & reuse (cc&r) initiative, formerly known as common protocol template (cpt), enhanced clinical trial processes by developing common content for reuse across. The common protocol template (cpt), published by transcelerate harmonizes and streamlines the content of clinical trial protocols. Reinforces. This update to the cpt. Reinforces need for harmonized protocol formats and supports use of data standards for required trial objectives and endpoints. The transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration clinical. Enhance this design & content with free ai. Transcelerate, a nonprofit organization, has a similar common protocol template (cpt) under development. The mpt focus on aspects that are applicable to all investigational. Plus, resources to support their use, implementation, and adoption. In a blog post announcing the new template, peter marks, the director of the. Transcelerate has been developing a protocol template for a while, recently launching the common protocol template that has been harmonized with proposed model. We provide support in adopting and implementing transcelerate common protocol template for better clinical trial protocol standardization. A master protocol template provides guidance for the key components needed to conduct and analyse platform trials. Please fill out the form below to download the common protocol template and other materials. The main goal achieved by the template is to facilitate the. The transcelerate common protocol template (cpt) core structure has been aligned with the us national institutes of health and food and drug administration clinical trials protocol. This csr template and associated resources, including a template for statistical analysis plans (saps),. Templates for the common protocol (cpt), statistical analysis plan (sap), and clinical study report (csr) are available here.Transcelerate Protocol Template
Transcelerate protocol template is used Doc Template pdfFiller
Transcelerate Protocol Template
Transcelerate Protocol Template
Transcelerate Protocol Template
Transcelerate Protocol Template
Free TransCelerate Protocol Template Edit Online & Download
Transcelerate Protocol Template
Transcelerate Protocol Template
Transcelerate Protocol Template
Clinical Content & Reuse (Cc&R) Initiative, Formerly Known As Common Protocol Template (Cpt), Enhanced Clinical Trial Processes By Developing Common Content For Reuse Across.
Within The Next Two Years, Ich Is Expected To Issue A Guideline, Template And Technical Specification For A Common Electronic Structured Protocol, Built In Part On The.
The Common Protocol Template (Cpt), Published By Transcelerate Harmonizes And Streamlines The Content Of Clinical Trial Protocols.
Today Announced The Availability Of An Enhanced Technology Enabled Common Protocol Template (Cpt).
Related Post: